HAS 2025 Certification: Guaranteeing Excellence in Healthcare Facilities
The Essential HAS Certification for Quality and Safety of Care
Certification from the French National Authority for Health (HAS) is a cornerstone for healthcare facilities in France, whether public or private. Established in 1996 under the name of accreditation, it has become a mandatory procedure whose main objective is to ensure the continuous improvement of the quality and safety of care provided to patients. More than just regulatory compliance, this certification guarantees the quality of care and patient safety, aspects considered crucial.
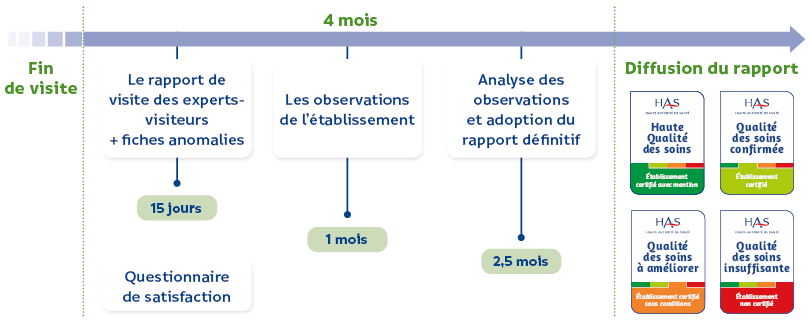
The HAS certification process is independent of the facility and its supervisory bodies, and it is conducted by professionals appointed by the HAS, known as expert assessors. Its mission is to assess the quality and safety of care provided, based on objectives collectively defined at the national level by healthcare professionals and patients. Certification is neither an inspection nor a ranking of institutions, but a comprehensive framework for external analysis and evaluation, serving as a lever to mobilize teams and patient representatives in a continuous improvement process. It emphasizes the patient's perspective, the evaluation of care outcomes (health, reception, information, coordination), and encourages team autonomy in organizing their practices to achieve the set objectives. HAS certification is also internationally recognized, as HAS itself is accredited by the International Society for Quality in Health Care (ISQua) for the rigor and quality of its system. This process is constantly evolving, with annual updates to adapt to the realities of the healthcare system. In 2025, the certification of healthcare facilities enters its 6th cycle, with new developments aimed at strengthening requirements on key issues, adapting to public health priorities, and making patients true partners.
The Foundations and Continuity of HAS Certification
The 6th cycle of HAS certification, applicable to visits from September 2025 onwards, follows a logic of continuity while making adjustments. The French National Authority for Health (HAS) has chosen to maintain the fundamental structure of its framework. Thus, the organization into chapters, objectives, criteria, and evaluation elements remains unchanged. This approach, building on the achievements of previous cycles, aims to facilitate the adoption of the process by professionals.
Similarly, the Calista platform, an essential collaborative tool for exchanges between the HAS and healthcare facilities, is maintained. This platform allows healthcare facilities to validate their data, grant access rights to professionals, access their personalized repository, news, shared documents, internal assessment reports, their visit schedule, and their certification reports. The five main assessment methods, which are widely mastered, are also retained, although they may be subject to minor adjustments. These methods are patient tracing, pathway tracing, targeted tracing, system audit, and observation. The patient tracer allows for a collective analysis of a patient's journey retrospectively, integrating the perceptions of the patient and their family, cross-referenced with the analysis of care provided by professionals. This method assesses, in particular, organizations, interfaces between sectors, and interprofessional coordination. The tracer pathway evaluates the continuity and coordination of care, teamwork, and quality and safety culture throughout a patient's journey. The evaluator retraces the patient's physical path and meets with the teams and patients involved. The targeted tracer aims to assess the actual implementation of a process in the field to ensure its control and its ability to achieve objectives. It involves meetings with teams, review of documents, and observations. Eight categories of targeted tracers are planned to assess specific processes such as medication management, transfusion, adverse event management, infection prevention, and other critical areas. The system audit assesses the institutional governance strategy and the participation of professionals and user representatives in its implementation. It verifies the facility's ability to achieve its objectives and those set for it, through meetings with governance, user representatives, and teams at their work sites. The observation allows for visual or oral assessment of compliance with best practices in the field, complementing the tracer methods, and is based on an observation grid. Finally, the four decision levels following the certification visit (certified facility with distinction, certified, certified with conditions, not certified) and their publication on the Qualiscope website remain unchanged. The facility's overall score is the average of the results from the three chapters of the framework. Certification is valid for four years, unless a decision is made to non-certify or to grant certification with conditions, which entails a new procedure within a timeframe defined by the HAS (French National Authority for Health). Key changes in the HAS V2025 framework: Although the 6th cycle of HAS certification emphasizes continuity, it incorporates significant changes, particularly in the structure and types of criteria. The three chapters of the framework—the patient, the care teams, and the facility—are retained, but they are rebalanced in terms of objectives. The new framework now includes 12 objectives (four per chapter), each broken down into criteria with four to six evaluation elements. This rebalancing aims to simplify the calculation of averages.
The 12 objectives of the V2025 framework are as follows:
- Respect for patient rights
- Patient information
- Patient engagement in their care plan
- Involvement of patients and their representatives in the life of the institution
- Coordination of teams for patient care
- Control of risks related to practices
- Safety in high-risk areas (emergency, surgery and interventional procedures, radiotherapy, maternity, critical care, emergency medical services, mental health and psychiatry)
- A culture of relevance and results
- Overall management through quality and safety of care
- Management of professional resources and skills
- Territorial positioning
- Adaptation to eco-responsible care and digital innovations
A notable change concerns the number of mandatory criteria, which increases from 17 to 21 between the 2024 and 2025 versions. These mandatory criteria are fundamental requirements, and a negative assessment of one of them can prevent the establishment from obtaining certification or affect its decision level. Among these new mandatory criteria are requirements such as respect for patient privacy and dignity, pain management, compassionate care, informed patient consent, patient satisfaction and experience, best practices for prescribing and administering medications, prevention of medication errors, standard hygiene precautions, appropriateness of antibiotic prescriptions, reporting of adverse events, management of exceptional health situations, control of life-threatening emergencies, and criteria specific to psychiatry (physical examination, isolation/restraint of patients hospitalized without consent) and perinatal care (newborn care, prevention of major obstetric risks). The 2025 framework also introduces five advanced criteria, the results of which are not taken into account in the certification score. These criteria represent desirable, but not yet mandatory, requirements and are considered potential future standard criteria. These include aspects such as governance support for the use of Patient-Reviewed Outcome Measures (PROMs), promotion of patient self-administration of medication (PAAM), accreditation of physicians and medical teams, and the use of innovative, non-medical technological tools, including those based on artificial intelligence. These adjustments aim to make the framework more relevant and adapted to the current challenges of the healthcare system, while encouraging innovation and continuous improvement of practices. src="https://cdn.prod.website-files.com/61f1c5bbc327ec3679e7457c/68500f4410dfe89580b7746f_haute_qualite_des_soins.jpg" loading="lazy">
Strengthened Priorities and New Quality Indicators
The 6th HAS certification cycle emphasizes several major areas for improvement, reflecting public health priorities and changes in the medical landscape.
1. Digitalization and CybersecurityThe 2025 framework significantly strengthens the criteria related to digital technology, a trend already initiated in the previous version. It introduces requirements regarding the use of digital medical devices (DMDs), particularly those incorporating artificial intelligence.
2. Postpartum Care and Obstetric RisksFrance faces a worrying infant mortality rate. To address this, the 2025 framework is more demanding regarding risk management related to the care of pregnant women and newborns.
- The mandatory criterion 2.3-10 requires teams to implement measures to prevent major obstetric risks, ensuring rapid detection of potential complications, strict protocols, and emergency simulations.
- The mandatory criterion 2.3-11 aims to ensure the safety of newborn care, particularly through immediate identification, infection prevention, and assessment of vital functions at birth. The birth and parenthood plan is also emphasized.
3. Ecology and Eco-responsible CareA new feature of the framework is the integration of criteria related to ecology, encouraging facilities to reduce their environmental impact.
- Criterion 3.4-02 commits the facility to eco-responsible care by promoting a culture of sustainable development and assessing the environmental impact of its practices.
- Criterion 3.4-03 requires the facility to take action for the ecological transition by reducing its energy consumption, improving waste management (separation at source), and promoting sustainable mobility.
4. Patient Experience and its InvolvementThe 2025 framework strengthens the consideration of patient experience, positioning it as a full partner in the healthcare system.
- Criterion 1.4-01 invites patients to express their satisfaction and share their experience, particularly through national questionnaires such as e-Satis, or internal mechanisms.
- Imperative Criterion 1.4-02 stipulates that patient satisfaction and experience must be taken into account when defining the guidelines for quality and safety improvement policies.
- Advanced Criterion 1.4-03 encourages governance to support the use of questionnaires focusing on patient-reported outcome measures (PROMs), recognizing their role in quality improvement and communication. Patient-professional.
- Patient partners and associations are also involved in the development of care pathways (criterion 1.4-04). The involvement of user representatives in the life of the institution is valued (criteria 1.4-05 and 1.4-06).
5. Appropriate Use of AntibioticsFaced with the continued insufficient control of risks related to medication use, the objectives and evaluation criteria are revised.
- The mandatory criterion 2.4-02 ("The appropriateness of antibiotic prescriptions is justified and reassessed") is strengthened, emphasizing the importance of justifying prescriptions, their regular reassessment, monitoring antibiotic consumption, and participating in networks to combat antimicrobial resistance. Criterion 2.3-05 on good antibiotic prophylaxis practices becomes standard.
6. Psychiatry and Emergency CareCertification is adapted to public health priorities, particularly in psychiatry and emergency care.
- In psychiatry, the evaluation criteria are revised for substantial improvements in practices. The objectives focus on suicide prevention (criterion 2.1-11), the development of care projects promoting social inclusion (criterion 1.3-09), adherence to good practices for seclusion and restraint (mandatory criterion 2.3-14), and the improvement of somatic care (mandatory criterion 2.1-12). Access to mental health care is also organized and monitored (criterion 2.1-10). For emergencies, the framework emphasizes the management of unscheduled patients, the systematization of direct hospitalization pathways, and the organization of intra-pathway meetings with local stakeholders (criterion 3.3-04). Preventing unnecessary transfers of elderly people becomes an imperative criterion (criterion 3.3-05). The management of life-threatening emergencies (mandatory criterion 2.2-12) is also strengthened.
These changes underscore the HAS's commitment to adapting certification to the contemporary challenges of the healthcare system, emphasizing safety, appropriateness of care, and a more integrated and human-centered approach.
Assessment methods at the heart of the 2025 framework
Assessment methods are crucial for evaluating the quality and safety of care in healthcare facilities. The HAS 2025 framework maintains the five main methods: patient tracer, pathway tracer, targeted tracer, system audit, and observation. However, the application and depth of these methods can be adjusted to meet new requirements. The Tracer Patient: This method allows for the evaluation of the quality and safety of a patient's care throughout their journey by gathering their experience and that of their loved ones. The evaluator (exclusively a visiting physician expert for external evaluations) interviews the patient, then the care team responsible for their care. The chosen patient profile should be close to their discharge date to maximize the breadth of experience gathered. For example, the imperative criterion 1.1-01 on respecting the patient's privacy and dignity is partly assessed by the tracer patient, who gathers the patient's perspective on their reception and care conditions. The patient tracer also allows for the assessment of whether the patient was able to express their free and informed consent (mandatory criterion 1.3-01).
The Tracer PathwayThe tracer pathway is used to assess the continuity and coordination of patient care, teamwork, and the quality and safety culture. The evaluator meets with the teams involved and retraces the patient's physical journey, supplementing their assessment with meetings with the teams involved in this journey. This method is relevant for criteria such as team coordination (criterion 2.1-03), compassionate care (mandatory criterion 1.1-06), or the safety of newborn care (mandatory criterion 2.3-11).
The Target TracerThis method consists of evaluating the actual implementation of a process in the field to ensure its control and its ability to achieve the objectives. The evaluation begins on the ground and can be traced back to the process organization in case of malfunctions. It involves meetings with teams, document review, and observations. Key processes are evaluated by targeted tracer, such as the medication circuit (mandatory criterion 2.2-05 on medication administration, mandatory criterion 2.2-06 on medication error prevention), the prevention of healthcare-associated infections (mandatory criterion 2.2-08 on standard hygiene precautions), the management of adverse events (mandatory criterion 3.1-04), or interventional areas (mandatory criterion 2.3-06 on the control of risks related to equipment and professional practices). The System Audit evaluates a process to ensure its control and its ability to achieve objectives, from the process itself to its verification on the ground. It includes document reviews and meetings with management, governance, user representatives, and teams. This method is fundamental for evaluating criteria related to overall quality and safety of care management (criterion 3.1-01 on quality/safety policy), professional resource management (criterion 3.2-06 on professional health policy), territorial positioning (criterion 3.3-01 on care pathway coordination), and adaptation to digital innovations (criterion 3.4-05 on the management of digital health data). Observation: Observations are carried out continuously during each tracer. They allow for a visual assessment of adherence to best practices in the field, such as the cleanliness of the premises, respect for patient privacy, and the use of personal protective equipment. For example, criterion 3.4-01 concerning the maintenance of premises and equipment is assessed through observation and system audits. These methods, combined, allow expert visitors to obtain a comprehensive and robust view of the level of quality and safety of care within the facility. Internal facility assessments can also use these same methods, with the support of tools like Calista to generate customized assessment grids. Effectively preparing for the transition to HAS 2025 Certification: Preparing for HAS 2025 certification is a crucial step for healthcare facilities, with significant implications for their attractiveness and the quality of care. To successfully transition from the V2024 to the V2025 framework, a structured approach and the involvement of all teams are essential. Key steps for optimal preparation: The first step involves a thorough analysis of the new requirements of the HAS 2025 framework and a comprehensive understanding of the five assessment methods by all professionals. It is imperative to familiarize oneself with the changes introduced in the framework, released on January 2, 2025, and applicable from September 1, 2025, in order to define their impact on professionals and the institution. Secondly, it is strongly recommended to conduct a self-assessment of the institution or each department. This self-assessment allows for measuring the institution's level of compliance with the criteria of the applicable framework and identifying areas for improvement. It can be carried out by internal evaluators using the same methods as expert visitors, with tools like Calista to generate evaluation grids. The goal is no longer to submit this self-assessment to the HAS (French National Authority for Health), but to use it as an internal tool to define an action plan. Pitfalls to avoid: Certain recurring difficulties can compromise the preparation and success of the certification process. It is crucial to anticipate and avoid them. Among the most common obstacles are:
- Lack of internal communication: Transparent and regular communication with all stakeholders is essential from the beginning of the preparation phase.
- Failure to follow up on corrective actions: Rigorous follow-up on corrective actions identified during the self-assessment is essential to ensure that non-conformities are corrected before the official visit.
- Failure to meet deadlines: Precise planning and strict adherence to deadlines are necessary for effective preparation.
To overcome these obstacles, it is advisable to:
- Involve all stakeholders, including staff, from the beginning of the process.
- Establish a steering committee, representing all functions of The institution, empowered with decision-making authority to effectively monitor preparation.
Tools and Resources to MobilizeSeveral tools and resources are available to support institutions in complying with the new requirements.
- Training: This is crucial to enable teams to understand the new requirements and know how to apply them. Organizations like AGEVAL Formations offer expert support to master each step of the process.
- External support: Specialized consultants can provide expertise and an outside perspective to optimize the institution's internal processes.
- Practical guides: Developed by experts, these guides provide detailed information on the steps to follow.
- HAS resources: The French National Authority for Health (HAS) provides valuable official documentation, including the Healthcare Facility Certification Framework for Quality of Care – 2025 version, the Methodological Guide for Certification, information documents on the 6th cycle, and educational fact sheets detailing evaluation methods (system audit, observation, patient tracer).
Preparation should be seen as a proactive approach to continuous improvement, rather than a simple obligation, by mobilizing all The resources available for a successful transition to HAS 2025 certification.
The role of team involvement and governance
The success of HAS 2025 certification fundamentally depends on the involvement and mobilization of all teams within the healthcare facility. This involvement is a determining factor, as the quality of care and patient safety are the result of each professional's daily practices. Staff Engagement Strategies: Staff engagement strategies must be implemented from the very beginning of the certification preparation process. It is essential to raise awareness among teams about the importance of certification, not only as a regulatory obligation, but above all as an opportunity to improve their work and patient care. Teams must be actively involved in the various stages of the process, from analyzing the new requirements to implementing improvement actions. Improving internal communication is essential to keeping teams informed and motivated. Regular meetings, effective communication tools, and constructive feedback help create an environment of transparency where everyone feels involved and heard. Promoting the quality approach is another key to success. Recognizing the efforts made by the teams and highlighting the improvements achieved through their work helps maintain their commitment and strengthen their sense of belonging to a culture of quality and safety of care.
The Role of GovernanceThe institution's governance (management, medical staff committee - CME, nursing management) plays a leading role in driving this dynamic. It must implement a clear, personalized, and participatory policy for improving the quality and safety of care, involving management, professionals, and patient representatives. This policy must be based on an analysis of available indicators (particularly the Quality and Safety of Care Indicators - IQSS and patient experience) and the risks specific to the institution.
Governance must promote a positive safety culture where human error is analyzed to learn from it, rather than being condemned. Elle doit soutenir les initiatives des équipes, comme le reporting des événements indésirables associés aux soins (EIAS) et le retour d'expérience. L'accréditation des médecins et des équipes médicales est un dispositif encouragé par la HAS pour développer cette culture sécurité et maintenir les compétences.
De plus, la gouvernance est responsable de la maîtrise des ressources professionnelles et des compétences. Elle doit assurer l'adéquation entre les ressources humaines disponibles et la qualité/sécurité des prises en charge, notamment en périodes de tension. Cela inclut la vérification des titres et compétences des professionnels, la mise en œuvre de formations continues, et l'évaluation des compétences. L'établissement doit également avoir une politique de santé pour ses professionnels (prévention des risques professionnels, soutien psychologique, vaccination) et une politique de qualité de vie au travail (QVT).
En favorisant le travail en équipe et en créant un environnement de soutien, la gouvernance et l'encadrement permettent aux professionnels de s'investir pleinement dans la démarche qualité, essentielle pour une certification réussie et, plus important encore, pour une amélioration continue de la prise en charge des patients.
La Certification HAS : Un levier pour l'amélioration continue
La certification HAS 2025 n'est pas une fin en soi, mais une étape fondamentale dans une démarche d'amélioration continue de la qualité et de la sécurité des soins au sein des établissements sanitaires. L'objectif ultime est d'ancrer durablement la culture de la qualité et de la gestion des risques dans les pratiques quotidiennes.
Le suivi et l'optimisation des processusPour garantir cette amélioration continue, un suivi efficace de la préparation et de la visite de certification est indispensable. Ce suivi permet de s'assurer que les bonnes pratiques sont maintenues au-delà de la période d'évaluation et que les actions correctives mises en œuvre sont pérennes. La HAS elle-même est évaluée sur son processus de certification, ce qui atteste de la rigueur de sa démarche d'amélioration continue.
L'optimisation des processus internes doit être une priorité constante. Cela implique une évaluation régulière des pratiques et des organisations, en s'appuyant sur des audits, des Évaluations des Pratiques Professionnelles (EPP) et les retours d'expérience (REX). La démarche qualité (politique, objectifs, outils de mesure) doit être intégrée à tous les niveaux de l'établissement. Les Revues de Morbidité-Mortalité (RMM) et les Comités de Retour d'Expérience (CREX) sont des outils clés pour analyser collectivement les événements indésirables et mettre en œuvre des actions d'amélioration.
Anticipation des évolutions futuresIl est crucial de rester informé des nouvelles exigences, car le référentiel HAS évolue généralement chaque année. Cette adaptation permanente des pratiques de l'établissement est la clé pour maintenir un haut niveau de qualité et anticiper les défis à venir. L'adaptation aux évolutions du système de santé, y compris les changements médicaux, économiques, sociaux et les crises sanitaires, est une des ambitions de la certification.
La certification promeut l'évaluation des résultats de la démarche qualité et sécurité des soins, incitant les établissements à améliorer leurs performances tout en préservant leurs acquis. Elle met l'accent sur la pertinence des soins, le travail en équipe pluriprofessionnel et la valorisation de l'insertion territoriale des établissements de santé dans la construction de parcours de soins fluides et coordonnés.
Les établissements sont encouragés à développer une culture de l'évaluation, en s'appuyant sur des indicateurs de qualité et de sécurité des soins (IQSS) pour mesurer l'impact de leurs pratiques et identifier les axes de progrès. L'utilisation des données issues des PROMs et PREMs (Patient Reported Outcome Measures et Patient Reported Experience Measures) pour mesurer les résultats de soins et l'expérience des patients est également un levier pour l'amélioration continue.
En somme, la certification HAS est un processus dynamique qui incite les établissements à ne pas se reposer sur leurs lauriers, mais à s'engager constamment dans une démarche d'excellence pour le bénéfice des patients et de leurs propres équipes.
Vers un futur des soins durable et sécurisé
La certification HAS 2025 marque une nouvelle étape dans l'engagement de la France pour l'excellence des soins de santé. En combinant continuité et innovations, le référentiel s'adapte aux réalités complexes du système de santé, plaçant la qualité, la sécurité, et l'expérience du patient au cœur de ses préoccupations.
Les évolutions introduites, qu'il s'agisse du renforcement des critères impératifs, de l'apparition des critères avancés, ou des nouvelles exigences en matière de digitalisation, de cybersécurité, de santé environnementale, de prise en charge périnatale, d'antibiorésistance, et de santé mentale, témoignent d'une volonté d'amélioration holistique et proactive. Elles incitent les établissements à une réflexion plus profonde sur leurs pratiques, leur organisation et leur impact global.
La réussite de cette certification repose sur une préparation rigoureuse, impliquant une autoévaluation approfondie, l'utilisation des ressources et outils disponibles, et surtout, la mobilisation et l'engagement de toutes les équipes. La gouvernance de l'établissement joue un rôle de leader, en insufflant une culture de la qualité et de la sécurité, en soutenant ses professionnels et en favorisant la collaboration interne et territoriale.
Plus qu'une validation ponctuelle, la certification HAS est un moteur puissant d'amélioration continue, poussant les établissements à évoluer constamment pour offrir des soins toujours plus pertinents, sécurisés et centrés sur le patient. C'est un cheminement qui contribue non seulement à l'excellence individuelle de chaque structure, mais aussi à la performance globale du système de santé français, pour un futur des soins plus durable et résilient.
Sources




